When a reaction reaches equilibrium, reactants are converted to products at the same rate that products are converted back to reactants. To maintain this balance, the reaction must occur in a closed system, which is a system where no substances can enter or leave the reaction container.
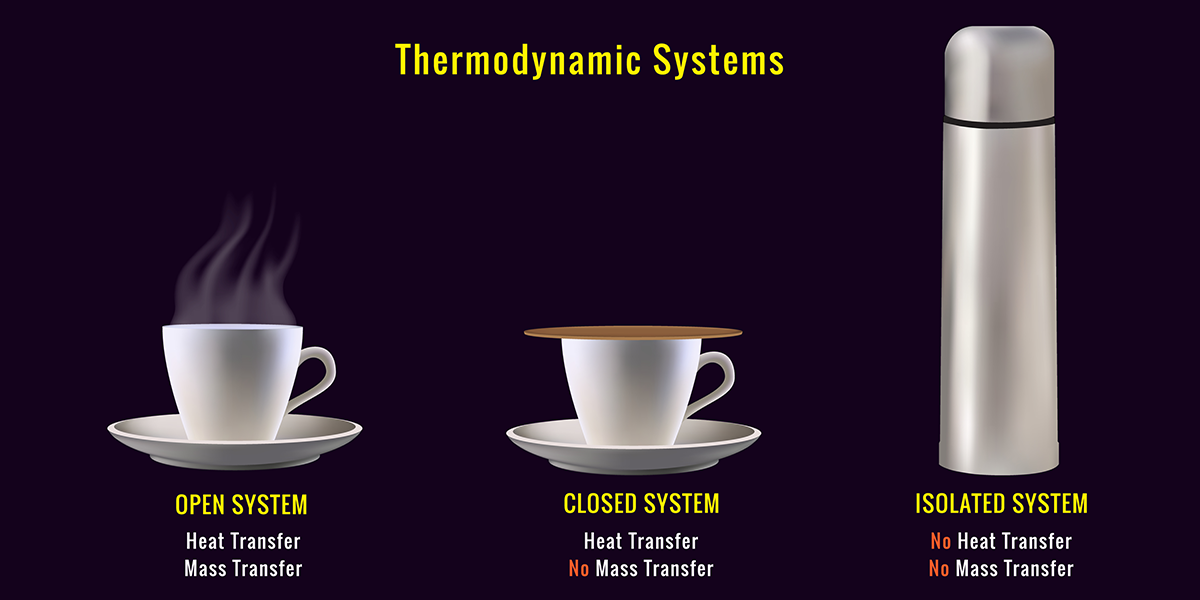
If the system is open to outside stresses, then the equilibrium can be disrupted. A stress is an outside influence that changes something about the reaction. Le Chatelier’s Principle states that if a stress is added to a system (i.e., a reaction) at equilibrium, then the system will shift the equilibrium to relieve the added stress.
Le Chatelier’s Principle in Simpler Terms
If something is added, the reaction will shift away to consume the excess that was added. If something is taken away, then the equilibrium will shift towards the void to replace what was taken.
A reaction at equilibrium can be affected by changes in concentration, temperature, and pressure. Le Chatelier’s principle predicts how these factors will influence the equilibrium position of a reaction.
Question
Why does the equilibrium constant (K) remain unchanged when concentration or pressure changes?
Because K depends only on temperature. Changes in concentration or pressure cause the reaction to shift to a new equilibrium position, but the constant ratio of products to reactants at that temperature stays the same.
