In this investigation you will be determining and comparing the specific heat capacities of 4 different materials, water, olive oil, brick, and iron.
The specific heat capacity of a substance is defined as the amount of energy it takes to change the temperature of 1 kg of that substance by 1°C. The specific heat capacity can be used to determine the amount of thermal energy that a substance absorbs or gives off (Q) as shown in the equation:
\({ Q = mc \Delta T }\)
In this equation, m is the mass of the material, c is the specific heat capacity of the material, and \({ \Delta t }\) is the change in temperature of the material.
In this lab you will be using this equation to calculate the specific heat. This can be done by rearranging the equation to:
\({ c = \frac{Q}{m \Delta T} }\)
Before you begin, click the Word or PDF link below to download your assignment worksheet. Then, read through the tabs below for the instructions. When you have completed the lab and the assignment, submit your worksheet to your teacher.
| Word |
Attribution: PhET Interactive Simulations | University of Colorado | https://phet.colorado.edu
The investigation consists of the parts shown below. Click the button below to download the instructions OR click through each tab to read and follow the steps to complete the investigation.
Calculating
Mass
Calculating Temperature Change
Calculating Specific Heat
For this investigation you will use the “Intro” mode of the PhET simulation.
When you first enter the “Intro” mode, you will see two blocks labeled iron and brick and two beakers, one with olive oil and one with water. To determine the specific heat of a substance, you must know its mass. Since a balance is not given in the simulation, you will use the density and the volume to find the mass, using the equation:
\({ \text{mass} = \text{density} \times \text{volume} }\)
-
First, find the volume of each liquid material (the water and the olive oil). The liquids are in beakers with lines on the outside. Each of the longer lines represent 1 L, and each of the smaller lines represents 0.2 L. Read the volume of each liquid material and record it in the data table of Question 1 on your assignment worksheet.
-
Convert the volume in L to cm3 by multiplying the volume in liters by 1000. Record the volume in cm3 in the data table of Question 1 on your assignment worksheet.
-
Next, find the volume of each solid material (brick and iron). Each side of the block of material is 10 cm. To find the volume of the blocks, use the equation for the volume of a cube:
\({ \text{volume} = \text{length} \times \text{width} \times \text{height} }\)
Calculate the volume in cm3 and enter it in the data table of Question 1 on your assignment worksheet.
- The density for each material is listed in the data table of Question 1 of your assignment worksheet. Use the density and the volume to calculate the mass with the equation given above. Then, enter it in the data table.
There are two heaters. To heat or cool the materials, you will put the material on the stand above the heater and then drag the slider up to heat the material and down to cool the material.
Calculate the temperature change of each material using the directions in this slideshow.
On the top left is a thermometer. The thermometer can be moved next to different materials to measure their temperature. Each line on the thermometer represents 25°C, so the thermometer shown here is measuring about 25°C. 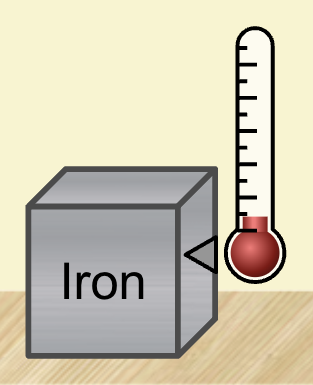
To find the temperature of a material, drag the thermometer to the right of the material with the arrow at the bottom facing the substance. When it is properly placed, the arrow will change colors to match the material as shown here. 
On the top right of the screen is a section with two check boxes. When “Energy Symbols” is checked, squares with an E in them are shown to represent the thermal energy of each material. The more energy symbols present, the more thermal energy a substance has. The energy symbols will move away from a material when energy is being given off and will go into the material when energy is being taken in. When “Link Heaters” is checked, this allows you to add/remove heat simultaneously for both materials. Now, you will find the temperature change when each material is heated. To make sure that each material receives the same amount of thermal energy, they will each be heated at the same time as water. When the water begins to steam, the temperatures will be recorded.
|
|
At this point you know the mass and the change in temperature for each material. You still need to know another variable before you can find the specific heats of the substances: the amount of thermal energy absorbed by each substance. To determine the amount of thermal energy absorbed, we will use the specific heat of water.
-
Calculate the thermal energy that the water absorbed, using the mass and change in temperature of the water according to the two data tables. Also use the specific heat of water which is 4.186 J/g°C. The equation for this calculation is:
\({ Q = mc \Delta T }\)
Show the work for this calculation on Question 3 on your assignment worksheet.
-
The amount of thermal energy that was absorbed by the water is equal to the amount of thermal energy that all the other materials absorbed. The thermal energy (Q) can then be used to find the specific heat of all the other materials using the equation:
\({ c = \frac{Q}{m \Delta T} }\)
Show the work needed to find the specific heat for each of the other materials on Question 4 on your assignment worksheet.
-
Using the specific heat capacity for each of the substances, answer Questions 5 and 6 on your assignment worksheet.

