
Have you ever noticed that it takes a long time for a swimming pool to warm up in the spring or that the climate at the beach is more moderate than it is inland? However, if you put a piece of metal outside, it warms up quickly. What's the difference? Adding a specific amount of energy (heat transfer) will change the temperature of different substances differently. It not only depends on the type of substance, but it also depends on the amount of the substance involved. Changing the temperature of a swimming pool will not take as long as changing the temperature of the ocean by the same amount!
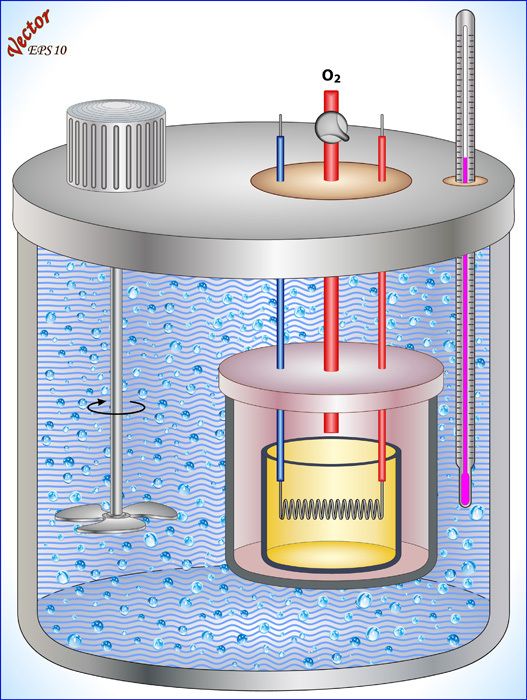
In science, we quantify this concept using a substance's specific heat capacity. The specific heat capacity of a particular substance is defined as the amount of energy it takes to change 1 kg of that substance by 1°C. Every substance has a unique specific heat capacity. Scientists have measured the specific heat capacity of these substances using calorimetry. A specially designed calorimeter is used to measure how much energy it takes to raise the temperature of a specific substance. It contains a thermometer, a stirrer, water at a specific temperature, and a test substance at a specific temperature.
Through repetitive tests, scientists have come up with the accepted specific heat capacities of many substances.
Question
Why do you think water takes a long time to warm up?
Water has a very high specific heat capacity. It takes a lot of energy to raise the temperature just one degree.
