Any nucleus that has more than 83 protons is too large and is considered unstable. If the nucleus of an atom is unstable, it will undergo radioactive decay until it is stable. Radioactive decay is usually denoted by a simple chemical equation where there is an unstable element on one side of the equation and then a more stable element on the other along with either an alpha, beta, or gamma particle.
\(\Large\mathsf{ ^{238}_{92}\text{U} \rightarrow ^{234}_{90}\text{Th} + ^4_2\text{He} }\)
Keep in mind that the total for the atomic number and mass number of the parent nucleus on the left must equal the sum of the atomic numbers and mass numbers of the daughter nucleus and the decay particle.
A radioactive atom may undergo three different types radioactive decay: alpha decay, beta decay, or gamma decay. Each has its own characteristics. Read through each tab below to learn about the defining characteristics of each type of decay.
Alpha Decay
Beta Decay
Gamma Decay
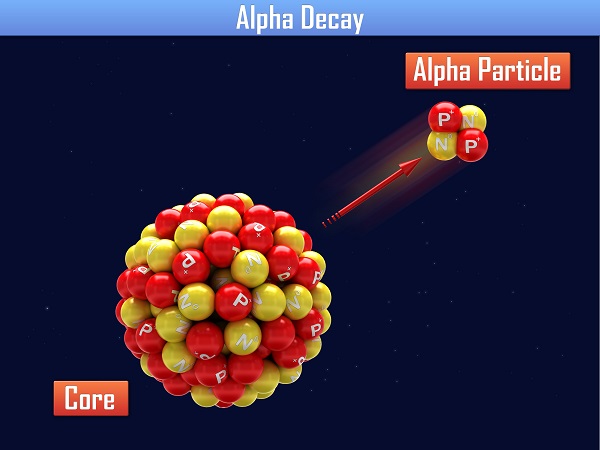
Nuclei that have more than 83 protons experience alpha decay to move toward the line of stability. Alpha decay specifically involves the release of a helium nucleus called the alpha particle. Alpha decay changes the mass of the nucleus because it loses two protons and two neutrons. The result is a lighter nucleus that has less positive charge than it had previous to the decay. An example alpha decay equation is that of uranium-238's decay into Thorium-234.
\(\mathsf{ ^{A}_{Z}\text{X} \rightarrow ^{A-4}_{Z-2}\text{X'} + ^4_2 \alpha }\)
There are actually two types of beta decay, often referred to as beta-plus and beta-minus. If an atom has too many neutrons, it will undergo beta-minus decay to change one of the neutrons into a proton. In beta-minus decay, the parent nucleus emits a high-energy electron.
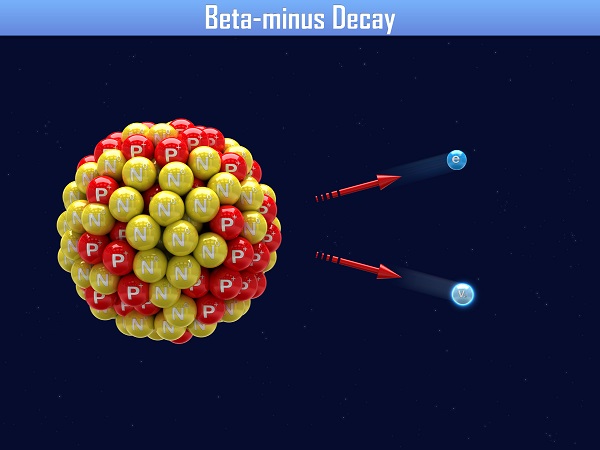
\(\mathsf{ ^{A}_{Z}\text{X} \rightarrow ^{A}_{Z+1}\text{X'} + ^0_{-1} \beta ^- }\)
If an atom has too few neutrons, it will undergo beta-plus decay to change one of the protons into a neutron. In beta-plus decay, the parent nucleus emits a positron. A positron has the same mass as an electron, and an equal but opposite charge. If a positron runs into an electron, they annihilate each other.
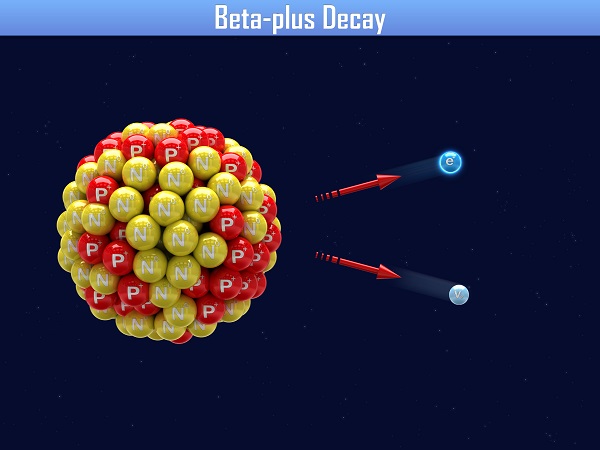
\(\mathsf{ ^{A}_{Z}\text{X} \rightarrow ^{A}_{Z-1}\text{X'} + ^0_{1} \beta ^+ }\)
Notice that there is another particle in the image for both beta-minus and beta-plus decay. In 1930, Wolfgang Pauli explained that in order for the momentum to be conserved in these decay reactions, a third particle called a neutrino had to exist. It wasn't until 1956 that the particle was actually discovered. In beta decay, a antineutrino is also released with an electron and an neutrino is also released with a positron.
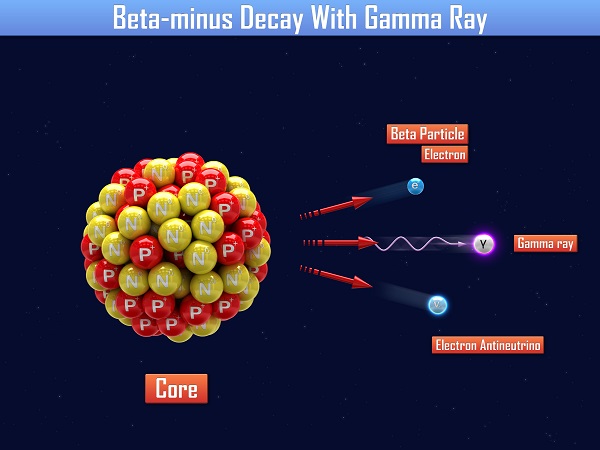
Gamma decay is the most complex, because it does not change the atomic mass or the atomic number of the parent, it only serves to reduce the energy of the nucleus. Gamma decay occurs when a parent nucleus is excited and then returns to a more relaxed state.
\(\mathsf{ ^{A}_{Z}\text{X*} \rightarrow ^{A}_{Z}\text{X} + \gamma }\)
Gamma most often occurs with beta decay, because beta decay leaves the daughter nucleus in an excited state. Similar to the photon emission of an electron moving from an excited state to a lower state, the nucleus of an atom releases a gamma ray. There are two steps in most beta decays, first the beta decay and then the gamma decay. For example, boron might decay to carbon in an excited state, then the excited carbon will release a gamma particle.
\(\mathsf{ ^{12}_{5}\text{B} \rightarrow ^{12}_{6}\text{C*} + ^0_{1} \beta^+ }\)
\(\mathsf{ ^{12}_{6}\text{C*} \rightarrow ^{12}_{6}\text{C} + \gamma }\)
