When a substance undergoes radioactive decay, the atom that decays is called the parent isotope. The atom that forms is called the daughter isotope. Not all isotopes decay at the same rate. Instead, each decays at its own distinctive rate. The table below shows you how these rates vary for a few select isotopes. The rate of radioactive decay is expressed in half-lives. Half-life is the amount of time it takes for one half of a parent isotope to decay to a daughter isotope. In other words, half the original atoms of the parent isotope decay to the daughter isotope in one half-life. From the table, you see that it takes 4.5 billion years for one-half of a uranium-235 sample to decay to its daughter isotope lead-206.
| Parent Isotope | Daughter Isotope | Half-Life (years) |
| Uranium - 235 | Lead - 206 | 4.5 billion |
| Potassium - 40 | Argon - 40 | 1.25 billion |
| Carbon - 14 | Nitrogen - 14 | 5,730 |
| Hydrogen - 3 | Helium - 3 | 12.3 |
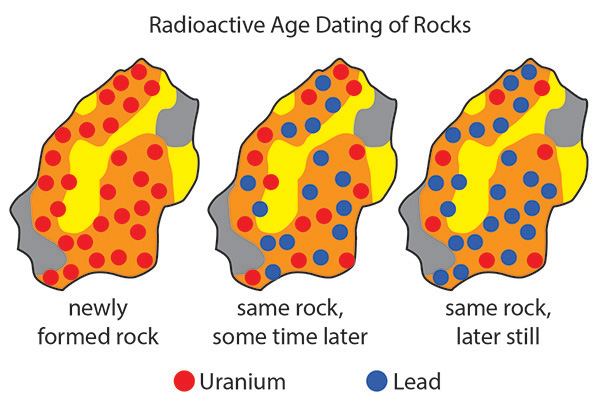
The concept of radioactive decay is crucial to absolute dating because it acts like a clock that records how much time has elapsed since a mineral within a rock was first formed. The sample you see here shows how the isotopes in a rock can change over time. Suppose a newly-formed rock contains a certain amount of the parent potassium-40 atoms, represented here by the red dots. Over time the potassium-40 isotopes decay to form argon-40 atoms, represented by the blue dots. The rate of that decay process is constant and the longer the time that passes, the greater the number of daughter atoms. Scientists can sample a rock and count the total number of parent and daughter atoms present. Using the proportion of parent to daughter atoms, they can determine how many half-lives have passed. They then multiply the length of the half-life by the number of half-lives that have passed to find the age of the rock.
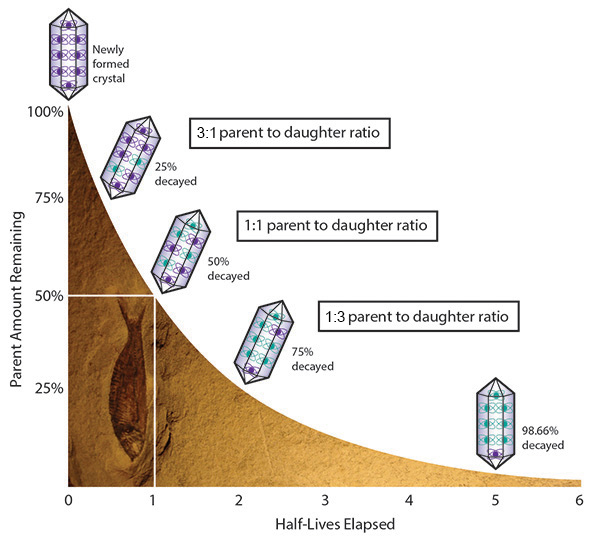
Suppose you find a fossil in a rock and want to know how old it is. You sample the rock and find that the proportion of parent atoms to daughter atoms is 1:3. This means you have 3 times more daughter atoms than parent atoms. It means that 75% of the parent atoms have decayed to daughter atoms. Following the graph, you see that this means that 2 half-lives have elapsed. If the half-life of your parent atom is 1 billion years, then you multiply 1 billion years times 2 to see that your rock is 2 billion years old. With tables of known half-lives, scientists have been able to assign ages to rocks found on earth.
Question
How long will it take for all of the parent isotope to decay?
Theoretically, this will never happen. Each half-life means that half will disappear and there is always half left. Of course, practically speaking, there will be a time that there are so few atoms left of the parent isotope that the effects are no longer seen.
