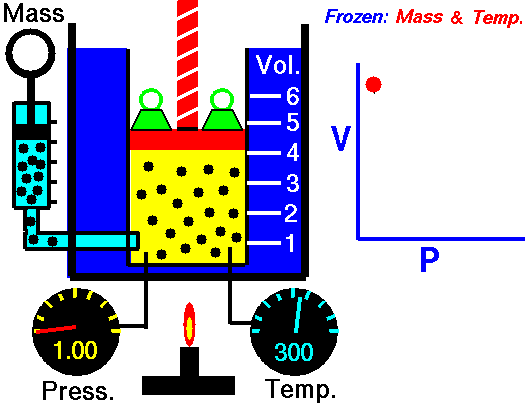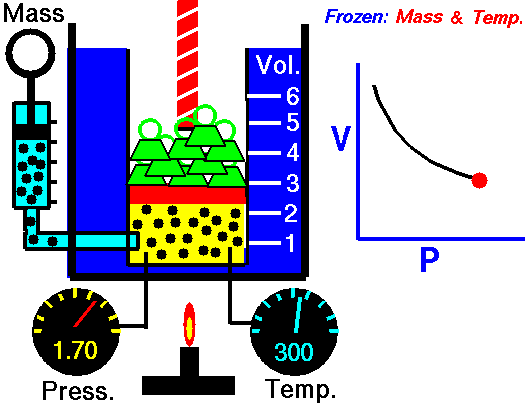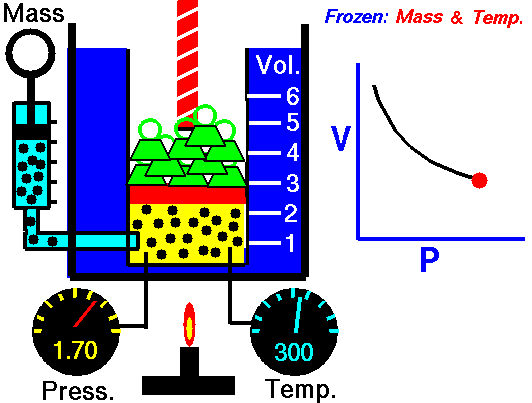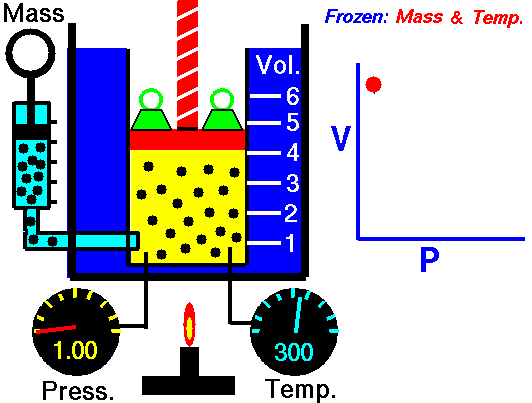
If you have a certain amount of gas (a certain number of molecules) and you keep the temperature the same, increasing the volume (measured by container size) will decrease the pressure, and decreasing the volume will increase the pressure. For a gas of a stable temperature, the pressure and volume of the gas are inversely proportional. What this means is that when you increase the volume of a gas, the pressure will decrease a corresponding amount. When you decrease the volume of the gas, the pressure will increase a corresponding amount. Any change in one will cause an inverse change in the other. An inverse relationship between two things means that if one gets bigger, the other gets smaller.
The amount of pressure impacts the solubility of gas. When a gas solute dissolves into a liquid solvent, it creates a solution. Generally, as pressure increases, the amount of a gas that is soluble in a liquid will also increase. Pressure affects the solubility of gases because if pressure on a gas is increased, more molecules of the gas will enter the liquid.
Pressure
If you have a certain amount of gas and you keep the temperature the same, what will decreasing the volume do to the pressure of the gas?
The pressure will increase.
