 A homogenous solution means
A homogenous solution means that the solution is the same throughout. The solution is mixed very well so each part of the liquid is the same as every other part. Mixtures can be homogeneous or heterogeneous (not the same throughout), but to be a solution, the mixture must be homogeneous.
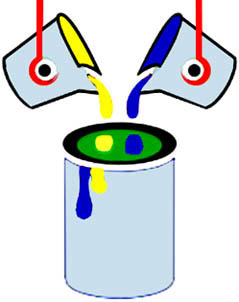
To understand the term “homogeneous,” think of mixing paints. When you first start to mix blue and yellow paints you can see bits of yellow paint, bits of blue paint, and bits of green paint. As you keep mixing though, eventually the paint becomes thoroughly mixed and the same throughout. Now all you see is green, and the mixture is now homogeneous. The blue and yellow have been mixed up completely to make the same green throughout.
In mixtures that are not solutions, the substances break down into smaller pieces but not to the size of molecules. Think of mixing clay with water. You can stir the clay around and around until you get a liquid. The clay has been broken down into smaller pieces than you started with, but the liquid is not clear. You can see little pieces of clay floating around. The clay has not been broken down into pieces the size of molecules and the mixture is not a solution.
 A homogenous solution means that the solution is the same throughout. The solution is mixed very well so each part of the liquid is the same as every other part. Mixtures can be homogeneous or heterogeneous (not the same throughout), but to be a solution, the mixture must be homogeneous.
A homogenous solution means that the solution is the same throughout. The solution is mixed very well so each part of the liquid is the same as every other part. Mixtures can be homogeneous or heterogeneous (not the same throughout), but to be a solution, the mixture must be homogeneous.