 If you dissolve one teaspoon of sugar or honey in a cup of tea, your drink becomes somewhat sweet. If it isn’t sweet enough for your taste, you add more sugar or honey to make it sweeter. As you add more sweetener, your solution becomes more concentrated. A concentrated solution has a large amount of solute dissolved in it. A dilute solution has a small amount of solute dissolved in it.
If you dissolve one teaspoon of sugar or honey in a cup of tea, your drink becomes somewhat sweet. If it isn’t sweet enough for your taste, you add more sugar or honey to make it sweeter. As you add more sweetener, your solution becomes more concentrated. A concentrated solution has a large amount of solute dissolved in it. A dilute solution has a small amount of solute dissolved in it.
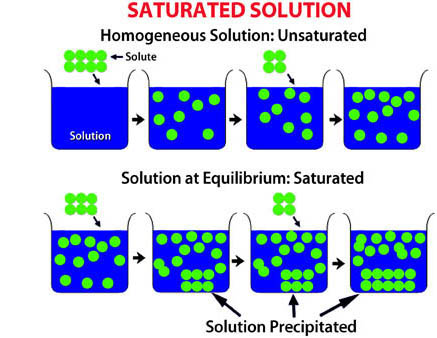
If you were to keep adding more and more sugar to the tea, would there ever be a point when you had added so much sugar that it could no longer dissolve? Yes! A solution which is so concentrated that it cannot dissolve any more of the solute is called saturated.
The word “saturation” is similar to the word “sate” (meaning satisfied); when you have eaten enough dinner and don’t want any more to eat, you are sated. A solution that can still dissolve solute is called unsaturated. As you have found, usually solutions that are warm or hot can dissolve more of the solute than solvents that are cool or cold. The amount of a solute that can be dissolved in a solvent is called its solubility.
Concentrated or Dilute
What does it mean to say a solution is more dilute?
A solution that is more dilute has a smaller amount of solute dissolved in it.
