 What would happen if you tried to use one in a flashlight? The electrolyte would spill out and the cell wouldn’t work. This is why dry cells are used instead.
What would happen if you tried to use one in a flashlight? The electrolyte would spill out and the cell wouldn’t work. This is why dry cells are used instead.
The precursor of the dry cell was invented in 1866 by George Leclanche of France. This was a huge improvement as now batteries were portable. Dry cells are not completely dry; instead of a liquid electrolyte, a moist electrolyte is used. Batteries that we use in flashlights are examples of dry cells.
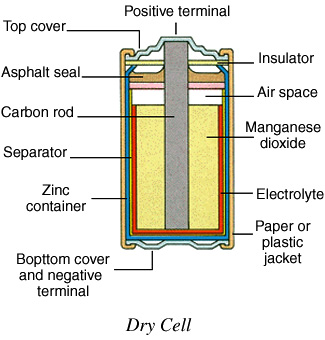
The 6-volt and D-cell size batteries we had you buy at the hardware store are examples of dry cell batteries. In a dry cell, the whole outside casing is made of zinc and is the negative electrode. On the batteries you are looking at, the outside of the zinc is covered with plastic to insulate you from shock. On the 6-volt battery part of the negative electrode sticks up at the end of the cell. It is marked with a symbol which looks like (–). This is called the negative terminal. On some dry cells there is a screw on the end so that you can wrap a wire around the terminal.
In the center of the zinc casing, inside where you can’t see it, is a carbon rod (carbon electrode). The positive terminal that sticks out of the 6-volt battery is attached at the top of the carbon electrode. It is marked with the symbol which looks like “+”. Now find the positive and negative terminals on the D-cell sized battery. As you can see, it is designed differently, but it works the same way.
Simplified diagram showing the layout of zinc-carbon type battery construction. Commonly called dry cells. The dry cell (shown above and at the right) works the same way as the wet cell. The moist electrolyte causes the electrons from the carbon to accumulate on the zinc, giving the zinc a negative charge. When the terminals are connected, the electrons move back to the carbon. Their movement causes an electric current.
Dry Cell Battery
What is the positive terminal connected to in a dry cell battery?
In a dry cell battery the positive terminal is connected to the Carbon Rod.
