 The electrolyte in the wet cell will continue to use its chemical energy to move the electrons from the copper to the zinc and the conductive wire will continue to let the electrons flow back to the copper. As long as the electrolyte acid has enough chemical energy, the wet cell will work, electricity will be generated, and the appliance will run. An example of this type of electric cell is a car battery.
The electrolyte in the wet cell will continue to use its chemical energy to move the electrons from the copper to the zinc and the conductive wire will continue to let the electrons flow back to the copper. As long as the electrolyte acid has enough chemical energy, the wet cell will work, electricity will be generated, and the appliance will run. An example of this type of electric cell is a car battery.
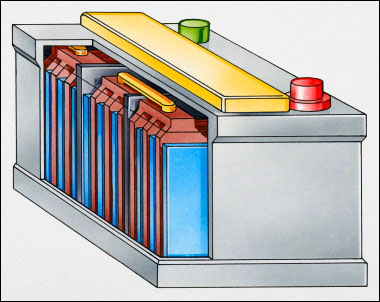
Car batteries are lead-acid batteries, which were invented in 1859 by Gaston Plant. A lead-acid battery is the most common battery used today. It can deliver a large amount of power (needed to start a vehicle), and can be recharged. When electrical current of the correct voltage is added to the battery, the chemical reaction is reversed, and the battery is recharged. Whenever you drive your car, the battery is being recharged. That’s why, if you left your lights on and the battery went dead, you need to let your engine run a while after you get a jump start from another engine. It is recharging your battery.
