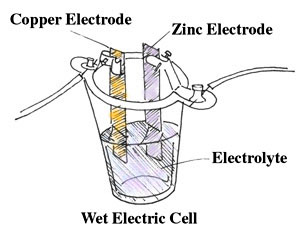
 A wet electric cell has all the components of the voltaic pile, but they are arranged a little differently. It contains two metals, zinc and copper, sitting in dilute sulfuric acid that touches both metals. The cell is called a wet cell because of the presence of the liquid acid, which is known as the electrolyte. The whole thing is sealed in an insulated case. Using chemical energy (energy that comes from chemical reactions - something you will learn more about later), the acid takes electrons from the copper and causes the electrons to collect on the zinc electrode. The zinc electrode now has “extra” electrons (which are negatively charged) making the zinc negatively charged.
A wet electric cell has all the components of the voltaic pile, but they are arranged a little differently. It contains two metals, zinc and copper, sitting in dilute sulfuric acid that touches both metals. The cell is called a wet cell because of the presence of the liquid acid, which is known as the electrolyte. The whole thing is sealed in an insulated case. Using chemical energy (energy that comes from chemical reactions - something you will learn more about later), the acid takes electrons from the copper and causes the electrons to collect on the zinc electrode. The zinc electrode now has “extra” electrons (which are negatively charged) making the zinc negatively charged.
What about the charge on the copper? Since it has fewer electrons than it should (because it gave some to the zinc), it is left with a proportionally higher number of protons (which are positive). The copper will have a positive charge.
In a wet cell battery, the electric current flows when a wire or other conductor connects the zinc electrode to the copper. The electrons will flow from the negatively-charged zinc back to the copper, as electrons would “prefer” to be equally balanced with protons. The flow of the electrons from the zinc to the copper causes an electric current.
Wet cell
How do electrons flow in a wet cell battery?
In a wet cell battery electrons flow from the zinc to the copper.
